What is a LiFePO4 Battery? Core Components & Chemistry
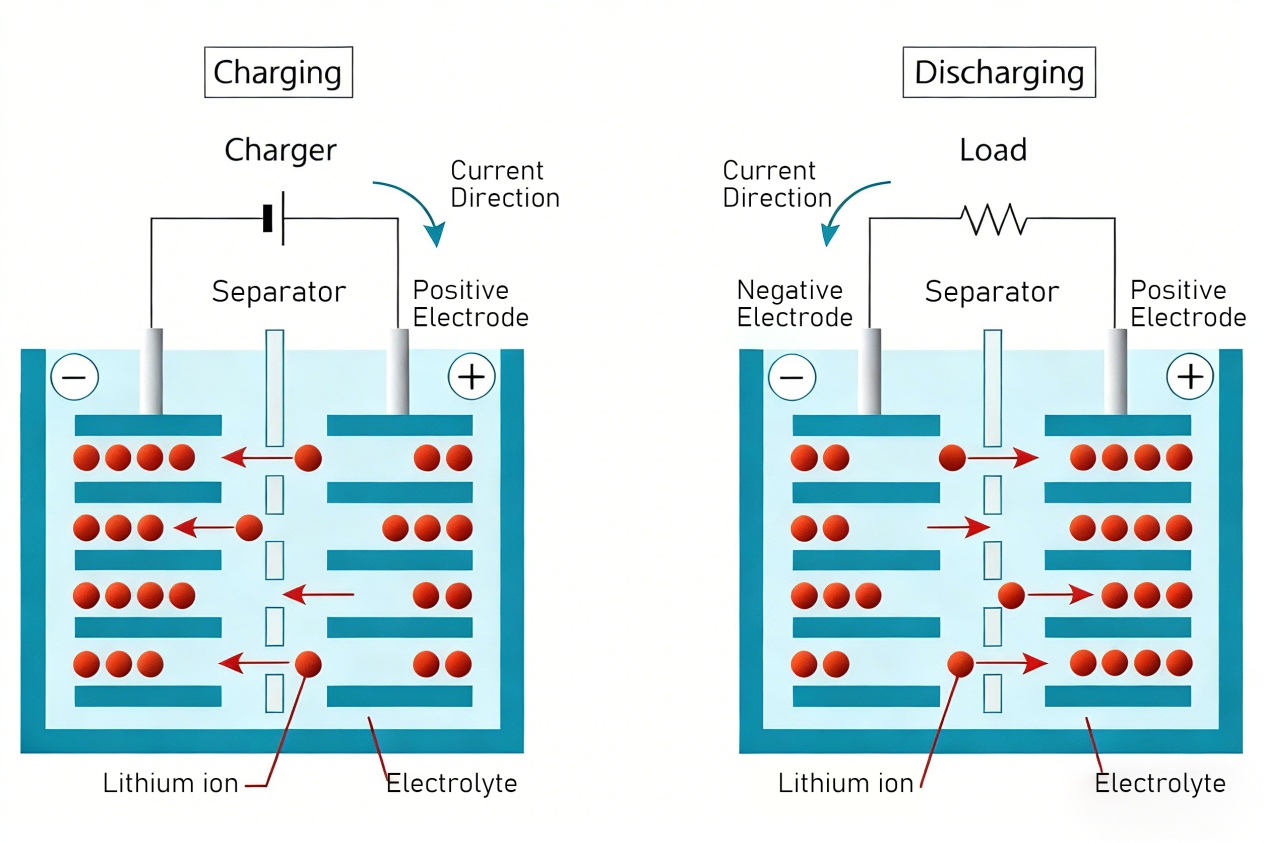
Lithium-ion batteries fall into two main categories: liquid lithium-ion batteries and polymer lithium-ion batteries. Liquid lithium-ion batteries are secondary batteries with Li+-intercalated compounds as electrodes. The positive electrode uses lithium compounds like lithium iron phosphate — the core material of LiFePO4 battery — LiCoO2, LiNiO2, or LiMn2O4. The negative electrode adopts lithium-carbon intercalation compound LixC6, and the electrolyte is an organic solution with lithium salts such as LiPF6 or LiAsF6. A typical system is LiC6|LiPF6-EC+DEC|LiCoO2. During discharge, lithium from the negative electrode dissolves into the electrolyte as ions, which then intercalate into the positive electrode crystal. During charging, this process reverses. Li+ shuttles back and forth between electrodes, earning it the name “Rocking Chair Batteries” — the foundation of LiFePO4 battery’s stable performance.
Classification of Lithium-Ion Batteries
- Lithium-ion batteries are generally liquid or solid-state, with polymer lithium-ion batteries belonging to the latter.
- In shape, they are typically cylindrical or prismatic. LiFePO4 rechargeable batteries, a versatile type of LiFePO4 battery, can also be customized into any shape, adapting to diverse applications like wearable devices and electric vehicles.
Internal Resistance
Battery internal resistance combines ohmic resistance from electrode materials, electrolyte, separator, and contact points and polarization resistance from electrochemical reactions. For LiFePO4 cells, low internal resistance is critical for high-current discharge — a key advantage of LiFePO4 battery in power scenarios.
Open-Circuit Voltage and Operating Voltage
- Open-circuit voltage is the potential difference with no external current, usually lower than the battery’s electromotive force. LiFePO4 batteries have a stable 3.2V open-circuit voltage across temperatures, ideal for steady-power use.
- Operating voltage, also called discharge or load voltage, is lower than open-circuit voltage due to internal resistance. It is influenced by discharge time, current, temperature, and cut-off voltage. Lithium iron phosphate batteries have a more stable operating voltage curve than LiCoO2 batteries, suiting devices needing consistent power.
Discharge-Related Parameters
- Discharge methods: Constant current, constant resistance, continuous, or intermittent discharge.
- Cut-off voltage: The minimum voltage to stop discharge — 2.5V for low-temperature or high-current, 3.0V for low-current — to extend LiFePO4 battery life.
- Discharge rate, also called C-rate: Expressed as hour rate or multiplier rate. For example, a 30Ah LiFePO4 battery discharged at 2A is 15-hour rate; a 3Ah battery at 2C uses 6A. The relationship is I = C/t — proper current control maximizes LiFePO4 battery performance.
Capacity and Specific Capacity
- Theoretical capacity: LiFePO4’s theoretical capacity is about 170 mAh/g, a reference for actual performance.
- Actual capacity: Commercial lithium iron phosphate batteries deliver 140–160 mAh/g, depending on production.
- Rated capacity: The minimum required capacity under specified conditions. Actual capacity is always lower than theoretical, with negative electrode capacity excess to ensure stable ion transfer for LiFePO4 battery.

Battery Life
- Secondary battery life includes cycle life (charge-discharge cycles) and wet storage life. LiFePO4 batteries offer 2000–5000 cycles versus 500–1000 for LiCoO2, a core advantage of LiFePO4 battery.
- Key influencing factors: Reduced active material surface area, shedding, corrosion, internal short circuits, separator damage, and crystal form changes. Controlling these ensures high-quality LiFePO4 battery.
Core Components of Lithium-Ion Batteries
Separator
Separators separate electrodes to prevent short circuits while enabling ion passage. For LiFePO4 batteries, they use organic solvent-resistant polyolefin porous membranes — PP, PE, or PE-PP-PE. PP stands for polypropylene and PE stands for polyethylene. Separator quality directly impacts LiFePO4 battery safety and durability, balancing ion permeability and mechanical strength.
Electrode Slurry Viscosity
Viscosity affects electrode quality — too low causes active material precipitation; too high hinders dispersion. For LiFePO4 slurry, controlling viscosity ensures uniform coating of high-density lithium iron phosphate powder, critical for consistent LiFePO4 battery performance.
Key Raw Materials
Composed of positive active materials like lithium iron phosphate and LiCoO2, negative active materials like graphite powder, copper foil as negative collector, aluminum foil as positive collector, separators, electrolytes (LiPF6 in organic solvents), and casings. High-purity lithium iron phosphate powder is vital for LiFePO4 battery capacity, cycle life, and safety.
The Complete LiFePO4 Battery Manufacturing Process
1. Slurry Preparation
Mix powdered positive (lithium iron phosphate) and negative (graphite) active materials with solutions and binders, stirring uniformly. Conductive agents like carbon black enhance conductivity between active materials and collectors. Uniform dispersion ensures consistent coating and LiFePO4 battery batch consistency.
2. Coating
Coat slurry on metal foils — aluminum for positive, copper for negative — and dry. Key parameters: Temperature, speed, and coating weight. Control coating weight by adjusting coater head gap based on slurry solid content and specific gravity. High-quality LiFePO4 electrode sheets are flat, uniformly coated, and free of defects.
3. Baking and Rolling
- Baking: Removes moisture and solvents from electrode sheets — moisture reacts with electrolyte to cause swelling.
- Rolling: Adjusts thickness to improve active material contact with collectors, reduce electron transfer distance, and increase capacity. Too thick reduces capacity; too thin wastes materials and limits high-current discharge for LiFePO4 battery.
4. Assembly
Clean sheets via brushing, weld tabs — ultrasonic for positive, resistance spot for negative — and stack or wind negative sheet, separator, positive sheet, separator to form a core. Tape core to prevent short circuits and deformation. Assemble in low-humidity environments to avoid moisture absorption, ensuring LiFePO4 battery safety.
5. Vacuum Baking and Electrolyte Injection
- Vacuum baking: Removes residual moisture and solvents from cores to protect electrolytes and electrodes.
- Electrolyte injection: Use manual or automatic injectors, then seal. LiFePO4 battery electrolyte, LiPF6 in EC or DEC, ensures high conductivity and compatibility.
6. Formation and Grading
- Formation: Activates materials via charge-discharge to form a stable solid electrolyte interface film, often called SEI film, improving LiFePO4 battery performance.
- Grading: Test and classify batteries by capacity to ensure compliance with rated standards.
Key Moisture Control in LiFePO4 Battery Production
Moisture is detrimental to LiFePO4 battery. Key measures: Implement moisture-proof protocols and shorten air exposure time; bake electrodes and cores; maintain low-humidity dry rooms for assembly and injection; regularly test moisture content in materials.
FAQs About LiFePO4 Battery
Key Q&As on lithium iron phosphate battery production and performance for industry pros and buyers:
Core factors:
1) Properties of active materials (purity and stability of lithium iron phosphate);
2) Impurities (residual moisture, metal contaminants) causing side reactions.
Key measures: Moisture-proof areas, shorten air exposure, bake components, and maintain low-humidity environments.
Lithium iron phosphate and graphite don’t dissolve in electrolyte — conductive agents enhance conductivity for efficient charge-discharge.
Measure slurry solid content and specific gravity, then adjust coater head gap to control thickness.
Flat surface, uniform coating, strong adhesion, dryness, and no defects (dust, scratches, bubbles).
Remove moisture and solvents — moisture causes swelling; solvents affect coating stability.
Too thick: Lower capacity and higher polarization. Too thin: Wasted materials and poor high-current discharge.
Remove moisture and solvents to protect components and ensure uniform electrolyte absorption.
Ready to Source High-Quality LiFePO4 Batteries?
Whether you need 50 custom batteries for a new robot model or over 1000 bulk units for your AGV fleet, we deliver industrial-grade LiFePO4 solutions tailored to your requirements.
- Bulk Procurement:Get a free bulk quote with worldwide shipping.
- Custom Design:Submit your robot/AGV specifications to receive a tailored battery solution.
Minimize downtime, cut replacement costs, and power your industrial operations with reliable LiFePO4 batteries.